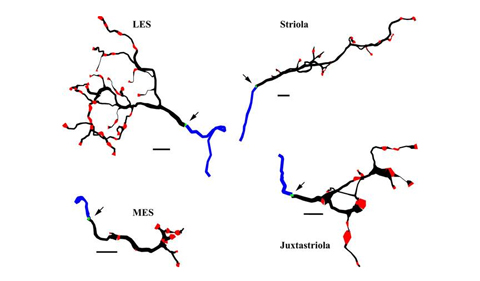
Terminal arbors of 4 afferents used in the simulations. Boutons are shown in red, the axon initial segment (AIS) in green, and myelin in blue. Clockwise from top left: lateral extrastriolar afferent (LES), afferent from the striola (zone 2), juxtastriolar afferent, and medial extrastriolar afferent (MES). The myelinated axons are truncated. As modeled, membrane area of the peripheral tree and average axon diameter are as follows: LES, 1,696 μm2 and 1.22 μm; striola, 1,786 μm2 and 1.85 μm; juxtastriolar, 1,157 μm2 and 1.7 μm; and MES, 502 μm2 and 1.23 μm. Scale bars, 10 μm. Arrows indicate location of the AIS (green) at the junction of the axon (black) and myelin (blue) segments.
The Journal of Neurophysiology gave a Facebook shout out to an article by Dr. William R. Holmes, Professor of Biological Sciences, and co-authors from the Biological Sciences neuroscience program.
“Using models of utricular bouton afferents, Ohio University researchers examined the role of afferent-hair cell connectivity in determining spike train regularity,” says the journal’s Facebook post.
The co-authors are
Abstract for Models of utricular bouton afferents: role of afferent-hair cell connectivity in determining spike train regularity: Vestibular bouton afferent terminals in turtle utricle can be categorized into four types depending on their location and terminal arbor structure: lateral extrastriolar (LES), striolar, juxtastriolar, and medial extrastriolar (MES). The terminal arbors of these afferents differ in surface area, total length, collecting area, number of boutons, number of bouton contacts per hair cell, and axon diameter (Huwe JA, Logan CJ, Williams B, Rowe MH, Peterson EH. J Neurophysiol 113: 2420–2433, 2015). To understand how differences in terminal morphology and the resulting hair cell inputs might affect afferent response properties, we modeled representative afferents from each region, using reconstructed bouton afferents. Collecting area and hair cell density were used to estimate hair cell-to-afferent convergence. Nonmorphological features were held constant to isolate effects of afferent structure and connectivity. The models suggest that all four bouton afferent types are electrotonically compact and that excitatory postsynaptic potentials are two to four times larger in MES afferents than in other afferents, making MES afferents more responsive to low input levels. The models also predict that MES and LES terminal structures permit higher spontaneous firing rates than those in striola and juxtastriola. We found that differences in spike train regularity are not a consequence of differences in peripheral terminal structure, per se, but that a higher proportion of multiple contacts between afferents and individual hair cells increases afferent firing irregularity. The prediction that afferents having primarily one bouton contact per hair cell will fire more regularly than afferents making multiple bouton contacts per hair cell has implications for spike train regularity in dimorphic and calyx afferents.
NEW & NOTEWORTHY Bouton afferents in different regions of turtle utricle have very different morphologies and afferent-hair cell connectivities. Highly detailed computational modeling provides insights into how morphology impacts excitability and also reveals a new explanation for spike train irregularity based on relative numbers of multiple bouton contacts per hair cell. This mechanism is independent of other proposed mechanisms for spike train irregularity based on ionic conductances and can explain irregularity in dimorphic units and calyx endings.

















Comments